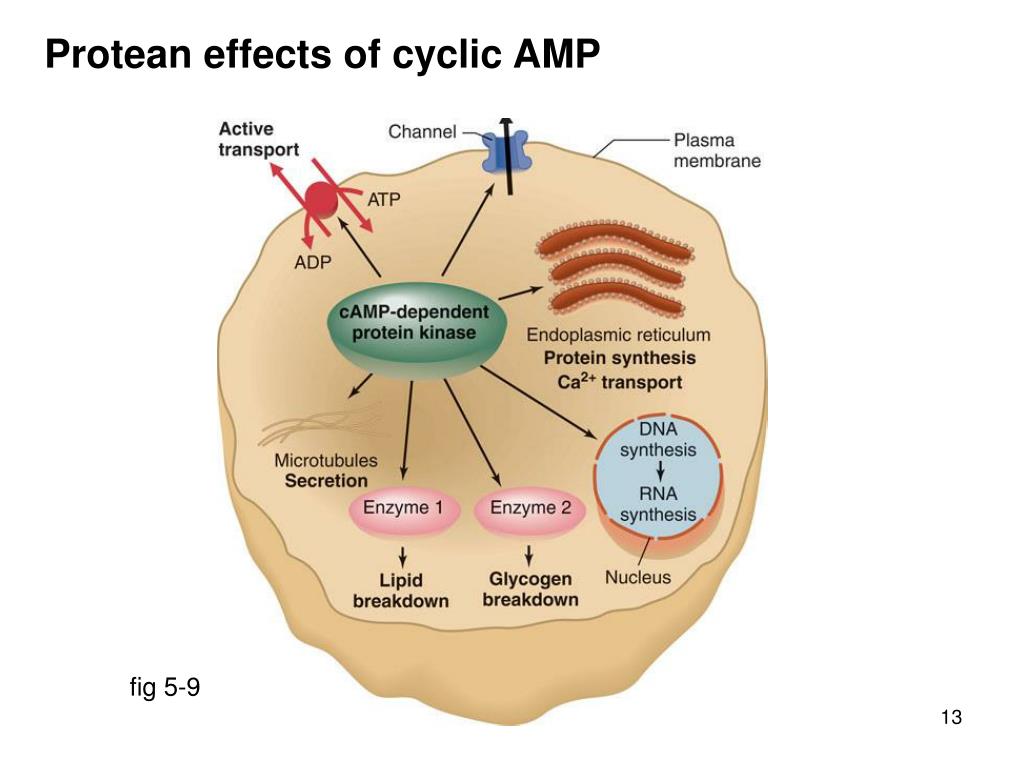
As the concentration of cyclic AMP increases, it binds to the regulatory subunits, leading to an allosteric change in conformation which causes unleashing of the catalytic subunits.When cyclic AMP levels are low, catalytic subunits are bound to a regulatory subunit dimer and are inactive.Intracellular concentration of cyclic AMP provides the most fundamental control over activity of protein kinase A: Type I enzymes inhabit cytoplasmic, soluble fractions of the cell, whereas type II enzymes tend to associate with cellular membranes. The different isotypes tend to have different distributions within cells and among tissues. In addition, three isotypes of the catalytic subunit have been identified (alpha, beta and gamma). Each of the four isotypes of the regulatory subunit are encoded by a different gene. Regulatory subunits exist in two major forms, RI and RII, with each form having two subtypes designated alpha and beta. Regulatory subunits may also have biologic activity distinct from their role in modulating catalytic subunit activity. This subunit also has has two domains that bind cyclic AMP, a domain that interacts with a catalytic subunit, and a "auto-inhibitory" domain that serves as a substrate or pseudosubstrate for the catalytic subunit. Regulatory subunit: Two molecules of this subunit bind one another in an anti-parallel orientation to form a homodimer for type I subunits (see below), this binding is covalent via disulfide bonds.It also contains a domain that binds ATP (the source of phosphate) and a domain that binds the regulatory subunit. Catalytic subunit: This subunit contains the enzyme's active site.The protein kinase A holoenzyme is a heterotetramer composed of two types of subunits: To put it another way, protein kinase A is ultimately responsible for essentially all of the cellular responses due to the cyclic AMP second messenger system. This enzyme thus functions as the end effector for a variety of hormones that work through a cyclic AMP signalling pathway. The unique characteristic of protein kinase A is that its activity is regulated by fluctuating levels of cyclic AMP within cells (hence its alias as the cyclic AMP-dependent protein kinase). DNA ligase is able to form a phosphodiester bond between the nucleotides.Like other protein kinases, protein kinase A (also known as the cyclic AMP-dependent protein kinase or A kinase) is an enzyme that covalently decorates proteins with phosphate groups. The relative ease of RNA hydrolysis is an effect of the presence of the 2' hydroxyl group.Ī phosphodiesterase is an enzyme that catalyzes the hydrolysis of phosphodiester bonds, for instance a bond in a molecule of cyclic AMP or cyclic GMP.Īn enzyme that plays an important role in the repair of oxidative DNA damage is the 3'-phosphodiesterase.ĭuring the replication of DNA, there is a hole between the phosphates in the backbone left by DNA polymerase I. The phosphodiester linkage between two ribonucleotides can be broken by alkaline hydrolysis, whereas the linkage between two deoxyribonucleotides is more stable under these conditions. Hydrolysis of phosphodiester bonds is catalyzed by phosphodiesterases, which are involved in repairing DNA sequences. In order for the phosphodiester bond to be formed and the nucleotides to be joined, the tri-phosphate or di-phosphate forms of the nucleotide building blocks are broken apart to give off energy required to drive the enzyme-catalyzed reaction. 
The negative charge attracts histones, metal cations such as magnesium, and polyamines. Repulsion between these negative charges influences the conformation of the polynucleic acids. Phosphodiesters are negatively charged at pH 7. 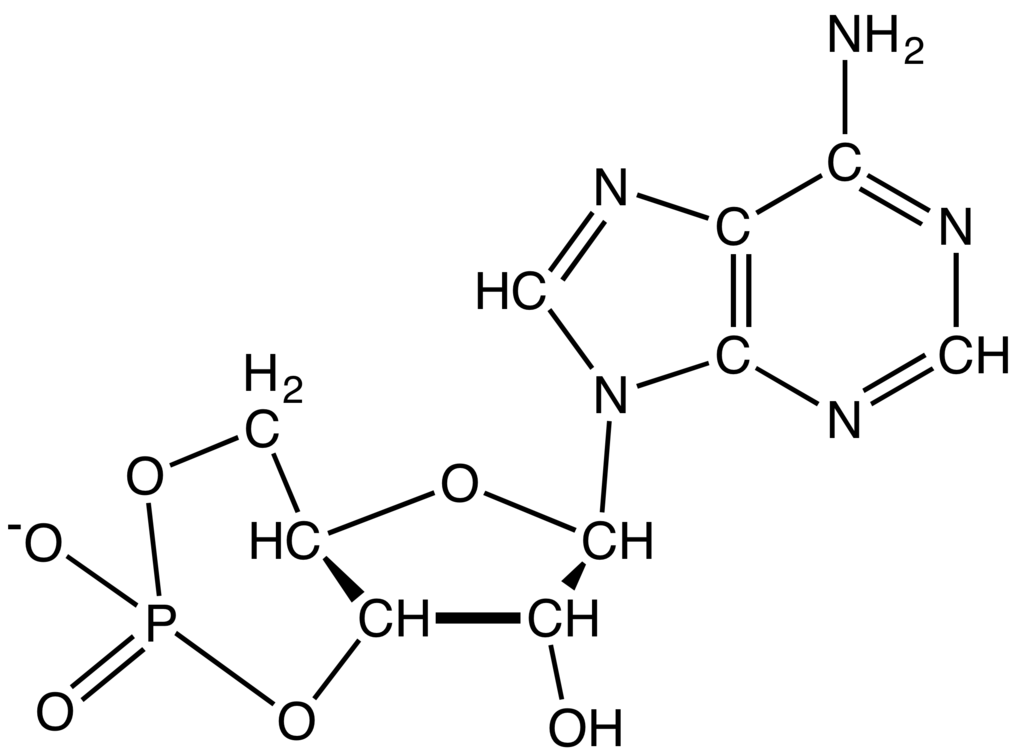
These saccharide groups are derived from deoxyribose in DNA and ribose in RNA. Specifically, the phosphodiester bond links the 3' carbon atom of one sugar molecule and the 5' carbon atom of another (hence the name, 3', 5' phosphodiester linkage ). The 3' carbon of one sugar is bonded to the 5' phosphate of the adjacent sugar. The phosphate is attached to the 5' carbon. Phosphodiester bonds make up the backbones of DNA and RNA. Discussion of phosphodiesters is dominated by their prevalence in DNA and RNA, but phosphodiesters occur in other biomolecules, e.g. The "bond" involves this linkage C−O−PO − 2O−C. In chemistry, a phosphodiester bond occurs when exactly two of the hydroxyl groups ( −OH) in phosphoric acid react with hydroxyl groups on other molecules to form two ester bonds. The 5' end has a 5' carbon attached to a phosphate, and the other end, the 3' end, has a 3' carbon attached to a hydroxyl group. –O– linkage between phosphoric acid and two other compounds Diagram of phosphodiester bonds ( PO 3− 4) between three nucleotides.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
